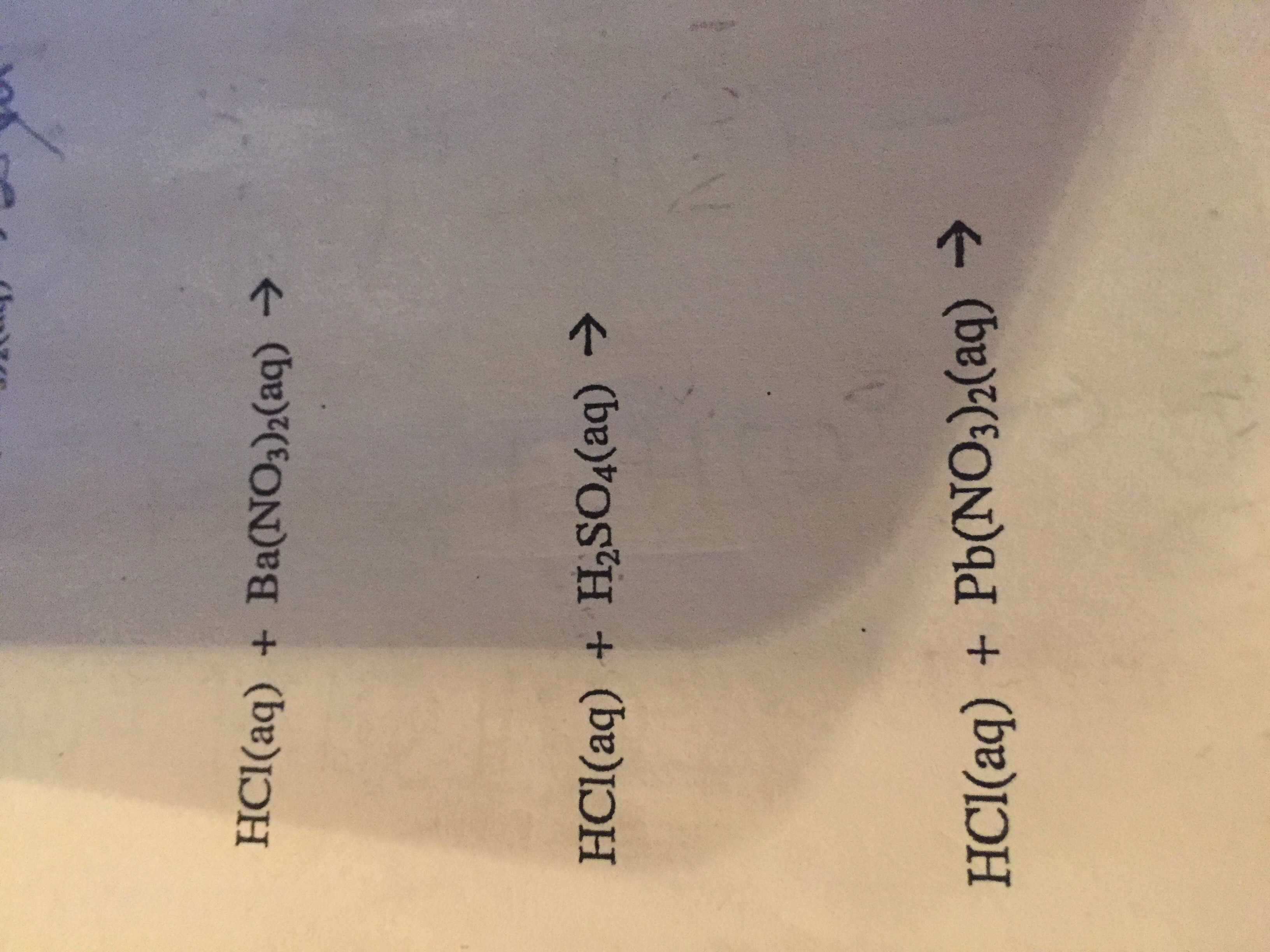
Aqueous barium nitrate reacts with sulfuric acid to yield a barium sulfate precipitate and nitric acid in - Brainly.in
which of the following pair will show common ion effect? (a)BaCl2+Ba(NO3)2 (b)AgCN+KCN (c)NaOH+NH4CN (d)HCOOH+H2SO4 GIVE REASON.EXPLAIN ALL OPTIONS
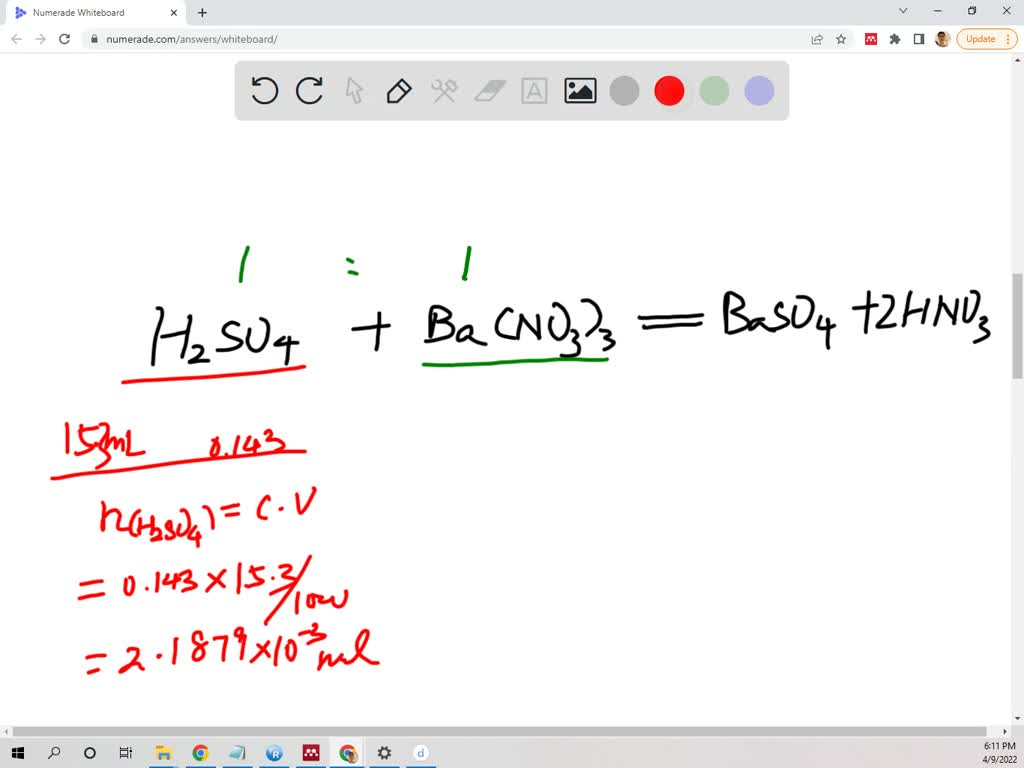
SOLVED: How many grams Ba(NO3)2 of are required to precipitate all the sulfate ion present in 15.3 mL of 0.143 M H2SO4 solution? Ba(NO3)2 + H2SO4 –> BaSO4 + 2HNO3
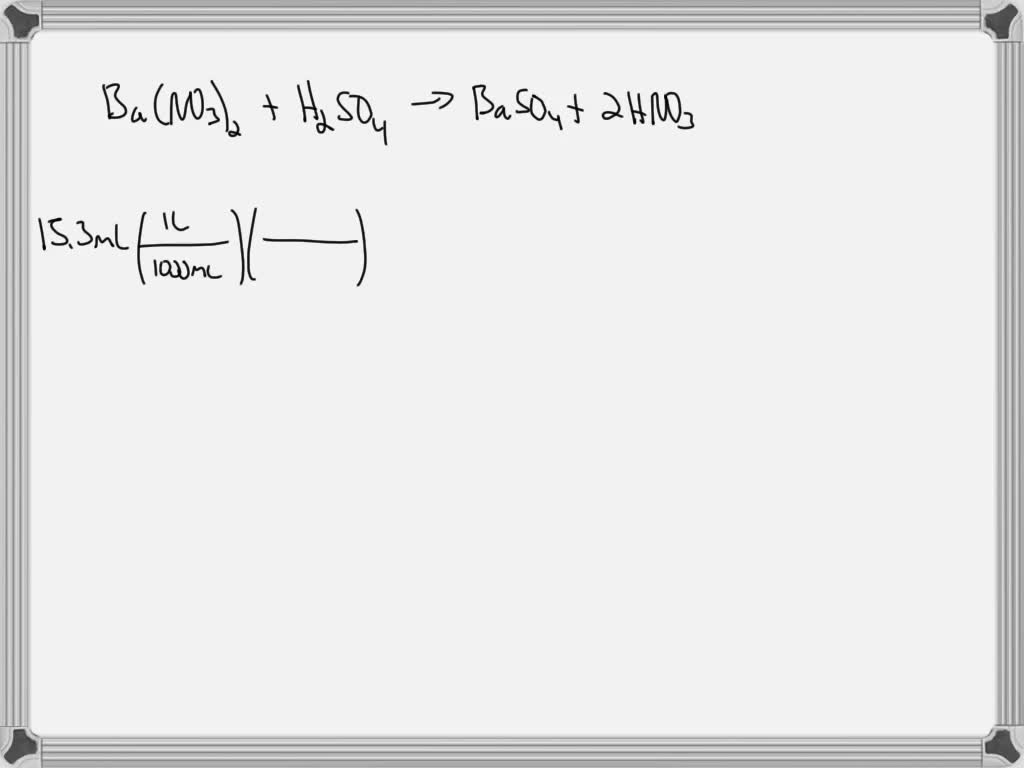
SOLVED: For the reaction Ba(NO3)2+H2SO4 =BaSO4+2HNO3 how many grams of Ba( NO3)2 are required to precipitate the sulfate ion present in 15.3 ml of 0.139 m of H2SO4

Chemistry: Chemical Word Equations Directions: Write a balanced chemical equation for each of the word equations below. - ppt download

How to Balance Ba(NO3)2 + Al2(SO4)3 = BaSO4 + Al(NO3)3 | Barium nitrate + Aluminum sulfate - YouTube