Spontaneity of the acid–base reaction between acetic acid and ammonia... | Download Scientific Diagram

Write the expression for the equilibrium constant, Kc for each of the following reactions.(i) 2NOCl (g) 2NO (g) + Cl2 (g) (ii) 2Cu (NO3)2 (s) 2CuO (s) + 4NO2 (g) + O2 (

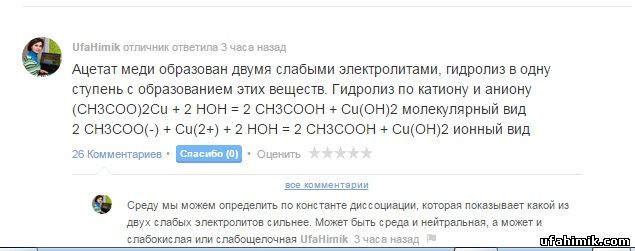
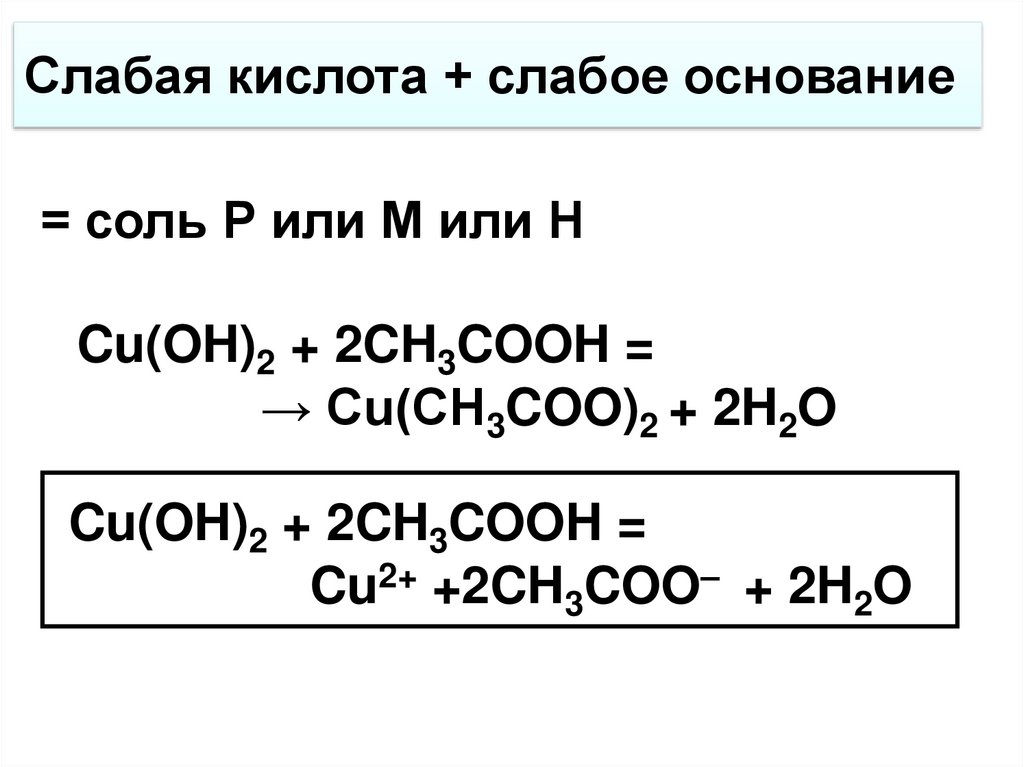
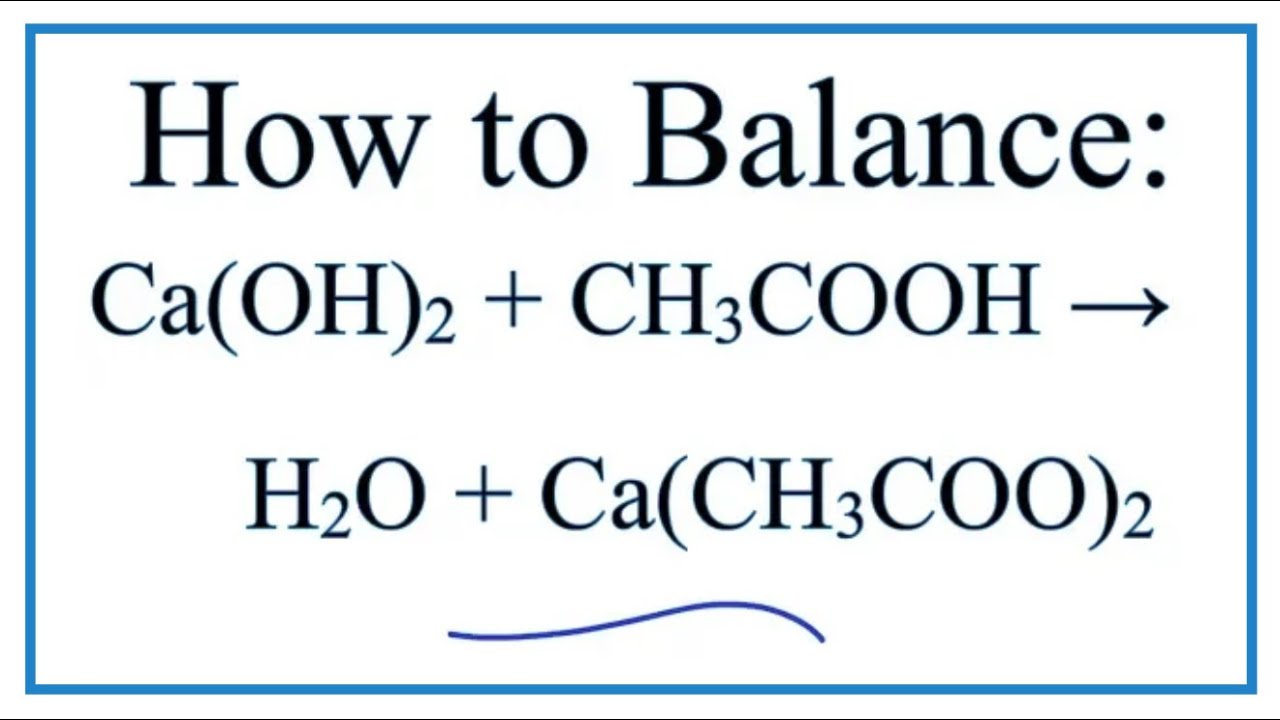
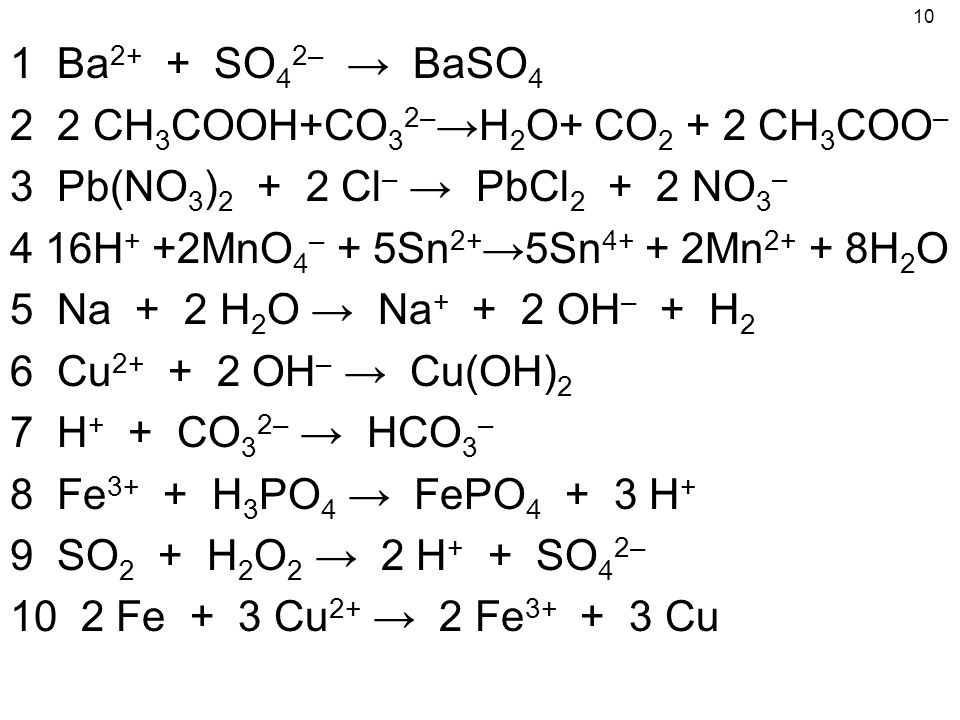
Помогите, пожалуйста! Вычислите Cu(OH)2 образующиеся при взаимодействии 10 грамм CuSO4 с - Школьные Знания.com

Write the expression for the equilibrium constant, Kc for each of the following reactions.(i) 2NOCl (g) 2NO (g) + Cl2 (g) (ii) 2Cu (NO3)2 (s) 2CuO (s) + 4NO2 (g) + O2 (

SOLVED: Answer the following questions using the following reactions: CH3COOH(aq) CH3COO (aq) Ht(aq) AH < 0 (1) Pb(OH)z(s) Pb*2(aq) ZOH (aq) AH > 0 (2) Cu(HzOJt2(aq) (Blue) ACI-(aq) Cucla ? (aq)(green) 4Hz0

Electrocatalytic Reduction of CO2 to Acetic Acid by a Molecular Manganese Corrole Complex - De - 2020 - Angewandte Chemie International Edition - Wiley Online Library

10 класс тема 7 дополнительное задание - 13 гдз по химии 10‐11 класс Радецкий дидактический материал

If CH(3)COOH+OH^(-)toCH(3)COO^(-)+H(2)O+q(1)and H^(+)+OH^(-)to H(2)O+q(2), then the enthalpy change for the reaction CH(3)COOHtoCH(3)COO^(-)+H^(+) is "equal to :